Process Validation Medical Device
Process Validation Medical Device in Accordance with 21 CFR 820.75 for Successful FDA Submissions
Process Validation Medical Device and other types of products require validation in accordance with 21 CFR 820.75 for successful FDA submissions.
Strand Products, Inc. may serve as an extension of your team to help wherever you are in the development life-cycle.

Process Validation Medical Device Inspector
Effective Process Validation Contributes Significantly to Assuring Product Quality
Effective process validation for medical device contributes significantly to assuring medical products, such as drugs, quality. The basic principle of quality assurance for the medical industry is that a product, such as drugs should be produced that is fit for its intended use.
Medical Device Manufacturing Process Validation
Medical device validation for product and processes which are not verifiable by examination of objective evidence to confirm customer specifications have been fulfilled. Strand Products offers FDA process validation guidance for medical devices. Medical device validation ensures particular requirements for a specific intended use can be fulfilled consistently.
Process Validation for Successful FDA Submission for Medical Device
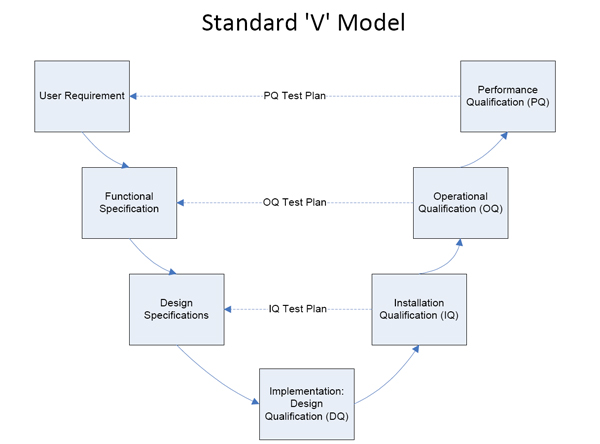
3 Steps of Process Validation Medical Device
Process validation for medical devices can be broken down into 3 steps:
- Process Design
- Process Qualification
- Continued Process Verification
Strand Products, Inc. offers complete medical device manufacturing validation support for the manufacture of your mechanical wire or stranded cable assembly. Medical device manufacturing process validation includes the following:
- Design Qualification (DQ)
- Install Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
Get Started Today with Medical Device Process Validation by Strand Products, Inc.
Strand Products, Inc. provides Medical Device Process Validation for Successful FDA Submission. Medical devices and other types of products require validation in accordance with 21 CFR 820.75 for successful FDA submissions. Strand is a leader in providing effective process validation.